The pre-release of the book 'Physics of Electrochemical Processes' (ISBN: 9789090332581) by P.M. Biesheuvel and J.E. Dykstra can now be downloaded from the website www.physicsofelectrochemicalprocesses.com free of charge.
Topics include:
- Ch. 1: The extended Frumkin isotherm describes the capacitive salt adsorption in intercalation materials
- Ch. 2: The Donnan model for the electrical double layer structure in charged materials, including electrodes
- Ch. 3: The Gouy-Chapman-Stern model and surface ionization
- Ch. 4: Volume effects in EDL theory (Bikerman, Carnahan-Starling, activity coefficients)
- Ch. 5: EDLs in motion: electrowetting, contact angle, energy harvesting
- Ch. 6: EDL interaction (DLVW theory)
- Ch. 7: Solute Transport (mass transfer to interfaces including dispersion)
- Ch. 8: Electrokinetics (hydrostatic and osmotic pressure, Navier-Stokes equation for electrolytes, osmosis vs electro-osmosis)
- Ch. 9: Heat effects for current flow across the EDL (Peltier effect, electrostatic cooling)
- Ch. 10: Acid-base reactions in transport models
- Preamble: The microscopic and experimental perspective of the electrical double layer
- Ch. 11: The difference between capacitive (non-Faradaic) and Faradaic electrode processes in electrochemistry
- Ch. 12: Electrode kinetics (in preparation)
- Ch. 13: Porous electrodes (in preparation)
- Ch. 14: Reverse Osmosis
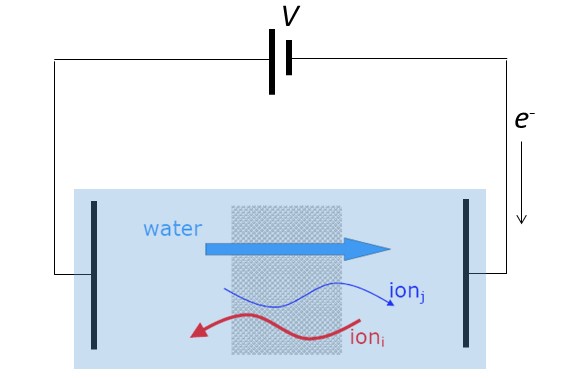
- Ch. 15: Electrodialysis
- Ch. 16: Ion transport in bio-electrochemical systems
- Ch. 17: Bioelectrochemical conversions on conductive media
- Ch. 18: Overview of electrochemical water desalination (in preparation)
- Ch. 19: Numerical methods (2021)
- Ch. 20: Analysis of experimental methods in electrochemistry (2021)